Biochemistry of molybdenum and tungsten cofactor biosynthesis
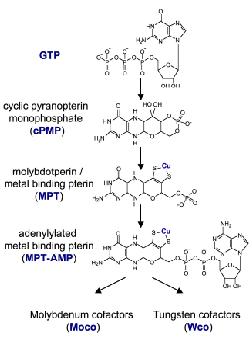
Molybdenum (Mo) and tungsten (W) enzymes catalyze important redox reactions in the global carbon, nitrogen, and sulfur cycles. Except in nitrogenases both metals are exclusively associated with a unique metal-binding pterin (MPT) that is synthesized by a conserved multistep biosynthetic pathway, which ends with the insertion and thereby biological activation of the respective element. Moco is synthesized by an ancient and conserved biosynthetic pathway that can be divided into four major steps, according to the biosynthetic intermediates cyclic pyranopterin monophosphate (cPMP), molybdopterin (MPT), and adenylated MPT (MPT-AMP). Different steps of the pathway have been studied in bacteria (E. coli), plants and humans. It is commonly believed that also tungsten-containing pterin cofactors mostly found in Archaea are synthesized in a similar way as Mo-cofactors with a specific mechanism for tungsten incorporation.
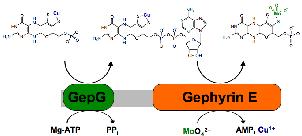
- Reaction mechanism of gephyrin-mediated molybdenum insertion in eukaryotes
We have uncovered the basic mechanism of molydenum insertion in eukaryotic Moco biosynthesis. Now we wish to understand the chemistry of the metal transfer reaction by studying the catalytiic proprties of the gephyrin, which combines the adenylylation and metal insertion function.
- Bis-molybdenum and tungsten cofactor biosynthesis in prokaryotes
The metal insertion reaction largely differs between pro- and eukaryotes resulting in the formation of mono- and bis-molybdenum-coordinated pterins with and without additional nucleotide modifications. In addition, many archaea express tungsten-containing enzymes that use the same type of pterin-based metal scaffold. Here we investigate the mechanism leading to metal specific cofactor formation bacteria and acheae.
- Function of copper in Moco synthesis
Copper was found to be bound to molybdopterin. We currently investigate the mechanism of copper transfer and release during the biosynthesis of Moco as well as its function for cofactor maturation. A putative link between molybdenum and copper homeostasis is of particular interest.
Funding

- DFG Schw 759/9-1 (2007-2010)
Tools
- purification of cPMP and in vitro conversion into molydopterin (MPT)
- recombinant protein expression in E. coli
- protein purification by affinity chromatography (IMAC, strep, etc.)
- protein crystallization and structure determination
- HPLC analysis of pterin derivatives
- enzyme assays and Mo enzyme assembly
- metal determinations and spectroscopy
Collaboration
- Prof. Dr. Graham George
University of Saskatchewan, Saskatoon, Saskatchewan, Canada - Prof. Dr. Fred Hagen & Dr. Peter-Leon Hagedoorn
Department of Biochemistry and Biotechnology, Delft University of Technology, The Netherlands - Prof. Dr. Ralf R. Mendel
Dr. Florian Bittner
Institute of Plant Biology, Technical University of Braunschweig, Germany
Project-relevant publications
- Bevers LE, Hagedoorn, Santamaria-Araujo JA, Magalon A, Hagen W,Schwarz G*
The function of MoaB proteins in the biosynthesis of molybdenum and tungsten cofactors.
Biochemistry (in press) - Llamas A, Tejada-Jimenez M, Gonzalez-Ballester D, Higuera JJ, Schwarz G, Galvan A, Fernandez E. 2007.
The Chlamydomonas CNX1E Reconstitute Molybdenum Cofactor Biosynthesis in E. coli Mutants.
Eukaryot Cell. 6:1063-67 - Fischer, K., Llamas, A., Tejada-Jimenez, M., Schader, N., Kuper, J., Ataya, F. S., Galvan, A., Mendel, R. R., Fernandez, E., and Schwarz, G. 2006.
Function and structure of the molybdenum cofactor carrier protein MCP from chlamydomonas reinhardtii.
J Biol Chem 281: 30186-94 - Llamas, A., Otte, T., Multhaup, G., Mendel, R. R., and Schwarz, G. 2006.
The Mechanism of nucleotide-assisted molybdenum insertion into molybdopterin. A novel route toward metal cofactor assembly.
J Biol Chem 281, 18343-18350. - Schwarz, G., and Mendel, R. R. 2006.
Molybdenum Cofactor Biosynthesis and Molybdenum Enzymes.
Annu Rev Plant Biol. 57: 623-647 - Schwarz, G. 2005.
Molybdenum cofactor biosynthesis and deficiency.
Cell Mol Life Sci 62, 2792-2810. - Llamas, A., Mendel. R.R., and Schwarz, G.* 2004
Synthesis of adenylated molybdopterin: an essential step for molybdenum insertion.
J. Biol. Chem. 279: 55241-55246 - Kuper, J., Llamas, A., Hecht, H.J., Mendel, R.R., and Schwarz, G.* 2004.
Structure of molybdopterin-bound Cnx1G domain links molybdenum and copper metabolism.
Nature 430: 803-806 - Guse, A., Stevenson,C.E.M., Kuper, J., Buchanan, G., Schwarz, G., Giordano, G., Magalon, A., Mendel, R.R., Lawson, D.M. and Palmer, T. 2003.
Biochemical and structural analysis of the molybdenum cofactor biosynthesis protein MobA.
J. Biol. Chem. 278: 25302-25307 - Kuper, J., Meyer zu Berstenhorst, S., Vodisch, B., Mendel, R.R., Schwarz, G.*, and Boxer, D.H. 2003.
In vivo detection of molybdate-binding proteins using a competition assay with ModE in Escherichia coli.
FEMS Microbiol. Lett. 218:187-193. - Kuper, J., Winking, J., Hecht, H.J., Mendel, R.R., and Schwarz, G.* 2003.
The active site of the molybdenum cofactor biosynthetic protein domain Cnx1G.
Arch. Biochem. Biophys. 411:36-46. - Hänzelmann, P., Schwarz, G., and Mendel, R.R. 2002.
Functionality of alternative splice forms of the first enzymes involved in human molybdenum cofactor biosynthesis.
J. Biol. Chem. 277:18303-18312. - Gutzke, G., Fischer, B., Mendel, R.R., and Schwarz, G.* 2001.
Thiocarboxylation of molybdopterin synthase provides evidence for the mechanism of dithiolene formation in metal-binding pterins.
J. Biol. Chem. 276:36268-36274. - Buchanan, G., Kuper, J., Mendel, R.R., Schwarz, G., and Palmer, T. 2001.
Characterization of the mob locus of Rhodobacter sphaeroides WS8: mobA is the only gene required for molybdopterin guanine dinucleotide synthesis.
Arch. Microbiol. 176:62-86. - Schwarz, G., Schulze, J., Bittner, F., Eilers, T., Kuper, J., Bollmann, G., Nerlich, A., Brinkmann, H., and Mendel, R.R. 2000.
The molybdenum cofactor biosynthetic protein Cnx1 complements molybdate- repairable mutants, transfers molybdenum to the metal binding pterin, and is associated with the cytoskeleton.
Plant Cell 12:2455-2472. - Kuper, J., Palmer, T., Mendel, R.R., and Schwarz, G.* 2000.
Mutations in the molybdenum cofactor biosynthetic protein Cnx1G from Arabidopsis thaliana define functions for molybdopterin binding, molybdenum insertion, and molybdenum cofactor stabilization.
Proc. Natl. Acad. Sci. U. S. A. 97:6475-6480. - Leimkühler, S., Angermüller, S., Schwarz, G., Mendel, R.R., and Klipp, W. 1999.
Activity of the molybdopterin-containing xanthine dehydrogenase of Rhodobacter capsulatus can be restored by high molybdenum concentrations in a moeA mutant defective in molybdenum cofactor biosynthesis.
J. Bacteriol. 181:5930-5939. - Witte, C.P., Igeno, M.I., Mendel, R., Schwarz, G., and Fernandez, E. 1998.
The Chlamydomonas reinhardtii MoCo carrier protein is multimeric and stabilizes molybdopterin cofactor in a molybdate charged form.
FEBS Lett. 431:205-209. - Schwarz, G., Boxer, D.H., and Mendel, R.R. 1997.
Molybdenum cofactor biosynthesis. The plant protein Cnx1 binds molybdopterin with high affinity.
J. Biol. Chem. 272:26811-26814.